본문
Mesoionic Carbene (MIC)-Catalyzed H/D Exchange at Formyl Groups
By Prof. Jean Bouffard (bouffard@ewha.ac.kr)
Department of Chemistry and Nanoscience
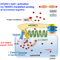
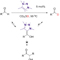
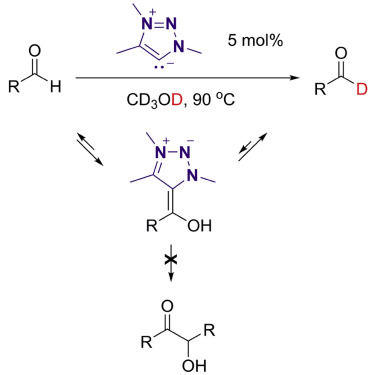
H/D exchange at formyl groups is the most direct approach for the synthesis of deuterated aldehydes. Platinum-group metal complexes have been employed to catalyze this transformation, with significant substrate scope limitations. Although N-heterocyclic carbenes can also activate the C–H bond of aldehydes through the formation of Breslow intermediates, benzoin condensation and other C–C-bond-forming pathways have so far outpaced synthetically useful H/D exchange. Investigation of the reaction of aldehydes with 1,2,3-triazolylidenes has revealed the reversible formation of Breslow intermediates and the inhibition of the condensation steps in methanol solvent. 1,2,3-Triazolylidenes catalyze the H/D exchange of aryl, alkenyl, and alkyl aldehydes in high yields and deuterium incorporation levels using deuterated methanol as an affordable D source. The unique properties of these mesoionic carbenes (MICs) enable a streamlined preparation of deuterated synthetic intermediates and pharmacophores that are highly valuable as mechanistic and metabolic probes.
* Related article
Wei Liu, Liang-Liang Zhao, Mohand Melaimi, Lei Cao, Xingyu Xu, Jean Bouffard*, Guy Bertrand*, Xiaoyu Yan*, Mesoionic Carbene (MIC)-Catalyzed H/D Exchange. Chem 2019, 5, 2484–2494.
https://doi.org/10.1016/j.chempr.2019.08.011