본문
Indoleamine 2,3-Dioxygenase-Expressing Aortic Plasmacytoid Dendritic Cells Protect against Atherosclerosis by Induction of Regulatory T Cells
by Prof. Goo Taeg Oh (gootaeg@ewha.ac.kr
)
Department of Life Science
Highlights
· Mouse and human aorta contain type I interferon-producing pDCs
· Selective depletion of aortic pDCs with BDCA2-DTR mice aggravates atherosclerosis
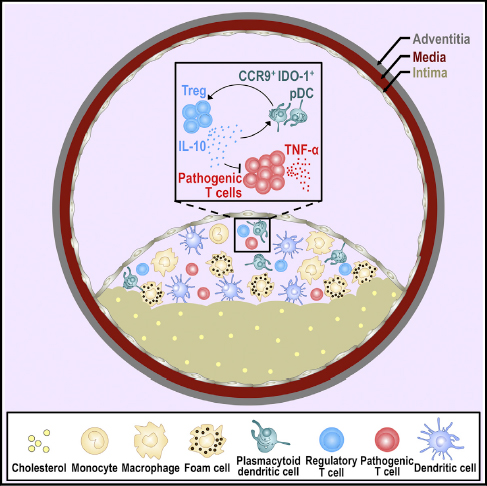
· pDCs and Tregs are concomitantly induced and colocalized in therosclerotic intima
· Local induction of Tregs by aortic IDO-1+ pDCs prevents atherosclerosis
Dendritic cells (DCs) are the principal cell type responsible for bridging innate and adaptive immunity and are central for the initiation of antigen-specific immunity and tolerance). DCs have been shown to reside in both lymphoid and non-lymphoid tissues. Recent studies on DC development and differentiation have defined subsets of DCs not only in lymphoid tissues but also in non-lymphoid tissues, including the aorta. The presence of DCs in the blood vessels of mice and humans suggests that these cells may be involved in atherosclerosis, a representative chronic vascular inflammatory disease. Interestingly, in healthy mice, DCs are mainly found in aortic roots, the lesser curvature of the aortic arch, and openings of arterial branches, regions with hemodynamic disturbance that promotes atherosclerosis in humans and experimental animals and that are also involved in lipid uptake and systemic lipid metabolism.
Plasmacytoid DCs (pDCs), although a rare bone-marrow (BM)-derived DC subset, serve an essential function by producing copious amounts of type I interferons (IFNs) upon microbial stimulation. Consequently, the potential function of pDCs in both innate and adaptive immune responses to most viruses has been the subject of intensive investigation. pDCs are also reported to have a role in peripheral and central tolerance, e.g., tolerance to vascularized cardiac allografts, tolerance against graft-versus-host disease, and chemokine (C-C motif) receptor 9 (CCR9)-dependent central tolerance. These tolerogenic properties of pDCs are mostly associated with the induction of regulatory T cells (Tregs). Importantly, systemic deletion of Tregs by anti-CD25 antibody (Ab) or selective depletion of Tregs by transgenic mice expressing diphtheria toxin receptor (DTR) under the control of Treg-specific Foxp3 promoter aggravated atherosclerosis. Several reports have begun to provide evidence, albeit conflicting, for pDCs’ role in mouse models of atherosclerosis. Although these previous studies are informative, they did not assess many classical functional features of pDCs in the aorta, including involvement in hematopoietic development, production of type I IFNs, localization in athero-prone areas of healthy and diseased aorta, and expression of known cell-surface markers specific for pDCs. Therefore, the identification of authentic pDCs in mouse and human aortas and the establishment of their contribution to the initiation, progression, or regression of atherosclerosis remain unmet challenges.
Addressing these gaps in our knowledge requires new approaches for identifying pDCs and assessing their role in atherosclerosis. To this end, we first sought to identify cells expressing pDC markers in healthy and atherosclerotic aortas of mouse and human. In this study, we show that these aortic cells are bona fide pDCs with the capacity to produce IFN-a. They also reside mainly in athero-prone areas of the healthy aorta but are distributed throughout lesions that develop in diseased aortas. Of note, aortic pDCs expressed indoleamine 2,3-dioxygenase (IDO-1), an enzyme critical to induce Tregs and Ido1 deficiency in athero-prone Ldlr_/_ mice had significantly decreased aortic Tregs. In line with this, when pDCs are selectively removed, mice developed more severe atherosclerosis, concomitant with a decrease in aortic Tregs. Finally, we also identified authentic tolerogenic pDCs in human atherosclerotic aortas and established their colocalization with Tregs.
On the basis of our findings, we propose that aortic pDCs described herein have two roles. First, they are part of the innate response initiated upon microbial stimulation (e.g., TLR7 and TLR9 ligation), a response concomitant with the rapid production of type I IFN. Confirming this role will require additional studies in vivo in the future. The second is that of an immune modulator. Accordingly, during atherogenesis, aortic pDCs increase in number and become one of the dominant cell types, thereby inducing Tregs and balancing the adverse effects of cytopathic T cells. Therefore, future therapeutic strategies harnessing tolerogenic pDC-based atheroprotective vaccines will be promising and beneficial for atherosclerosis in humans.
* Related Article
Tae Jin Yun, Jun Seong Lee, Kawthar Machmach, Dahee Shim, Junhee Choi, Young Jin Wi, Hyung Seok Jang, In-Hyuk Jung, Kyeongdae Kim, Won Kee Yoon, Mohammad Alam Miah, Bin Li, Jinsam Chang, Mariana G. Bego, Tram N.Q. Pham, Jakob Loschko, Jorg Hermann Fritz, Anne B. Krug, Seung-Pyo Lee, Tibor Keler, Jean V. Guimond, Elie Haddad, Eric A. Cohen, Martin G. Sirois, Ismail El-Hamamsy, Marco Colonna, Goo Taeg Oh*, Jae-Hoon Choi*, Cheolho Cheong*. Indoleamine 2,3-Dioxygenase-Expressing Aortic Plasmacytoid Dendritic Cells Protect against Atherosclerosis by Induction of Regulatory T Cells. Cell Metabolism. May 10;23(5):852-866, 2016.
Jae-Hoon Choi, Cheolho Cheong, Durga B Dandamudi, Chae Gyu Park, Anthony Rodriguez, Saurabh Mehandru, Klara Velinzon, In-Hyuk Jung, Ji-Young Yoo, Goo Taeg Oh* and Ralph M. Steinman*. Flt3 signaling-dependent dendritic cells protect against atherosclerosis. Immunity Nov 23;35(5):819-831, 2011.