본문
Environmental behavior of engineered nanomaterials in porous media: a review
by Prof. Ahjeong Son (ason@ewha.ac.kr)
Department of Environmental Science and Engineering
* In collaboration with University of South Carolina (Drs. Chang Min Park and Yeomin Yoon)
Nanoscience and nanotechnology are the fields that have become a significant scientific priority in research and development. They exploit unique nanoscale properties and phenomena of nanomaterials, which are now used in engineering. Nanomaterials are the materials having a domain size of ∼100 nm or less, and may resemble complexes of protein molecules. However, they differ from protein molecules due to their distinct structural features (e.g., mechanical, electrical, optical, magnetic, and biological properties), chemical composition, shape, size, density, aggregation, and types of surface. Unlike metal salts in solution, the main components of nanoparticles are normally not ionized; their oxidation state is often zero. However, it may be expected that the physicochemical properties of nanomaterials would differ substantially from those of bulk materials.
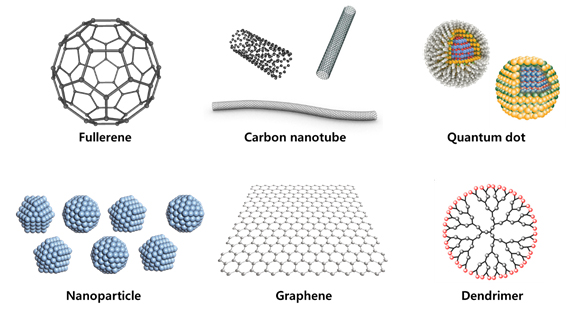
Figure. Illustration of nanomaterials and engineered nanomaterials
Engineered nanomaterials (ENMs) can be homogeneous or heterogeneous; these include particles with core–shell structures in terms of structure and composition, or multi-functional nanoparticles such as ‘smart’ nanoparticles developed for medical purposes. They have various structures, such as spheres, needles, tubes, and plates, with differing toxic effects in the human body and properties that can respond uniquely to biology or ecology. Due to their small sizes, nanometer-scale materials may pass into cells and translocate through or between cells. Electronic configurations and large surface-to-volume ratios, relative to bulk materials, result in increased particle surface energy with more biologically reactive sites as well as the structures per se.
The rapid proliferation of nanoparticle use has resulted in exposure to humans through environmental water, air, and soil systems. Environmental exposure to the most common engineered nanomaterials (ENMs), such as carbon-based and metal-based nanomaterials, can occur directly via intentional injection for remediation purposes, release during the use of nanomaterial-containing consumer goods, or indirectly via different routes. The nanoparticle behavior depends strongly on particle-specific properties, solution chemistry, soil hydraulic properties, and soil matrix properties when ENMs are released into the soil environment. Such factors are of importance in controlling the transport process of ENMs and their interaction with heavy metal ions. Thus, it is important to consider use and potential discharge of ENMs into soil environments and to determine the effects of environmental conditions on the behavior of ENMs in aqueous porous media.
Recent review articles have mostly covered risk assessment, toxicity, and life cycle analyses regarding the emission of ENMs. Some have also described occurrence, biotransformation of, and interaction between nanoparticles and biological systems using computational modeling techniques such as molecular dynamic methods. In this review, inevitable release of ENMs and their environmental behaviors in aqueous porous media are discussed with an emphasis on influencing factors, including the physicochemical properties of ENMs, solution chemistry, soil hydraulic properties, and soil matrices. Major findings of laboratory column studies and numerical approaches for the transport of ENMs are addressed, and studies on the interaction between ENMs and heavy metal ions in aqueous soil environments are examined. Future research is also presented with specific research directions and outlooks.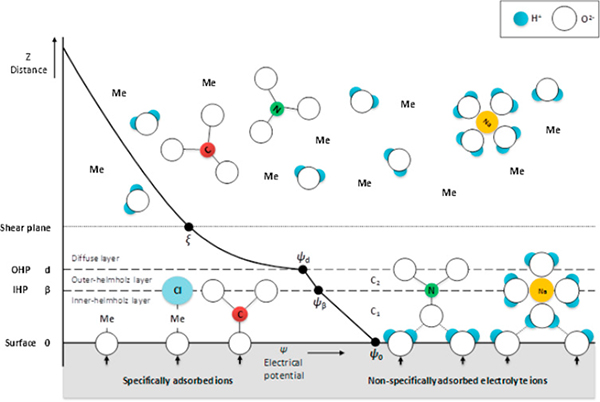
Figure. Schematic diagram of the structure of triple layer model and the charge distribution at a solid–liquid interface representing the placement of ions in the electrostatic plane.
* Related article and site
Chang Min Park, Kyoung Hoon Chu, Jiyong Heo, Her Namguk, Jang Min, Ahjeong Son, Yeomin Yoon*, (2016) Environmental behavior of engineered nanomaterials in porous media: a review. Journal of Hazardous Materials 309, 133-150